|
These all react with cold water with increasing vigour to give the metal hydroxide and hydrogen. This is because the metal hydroxides thermally decompose (split up on heating) to give the oxide and water. If it reacts with steam, the metal oxide is formed. Note: As a general rule, if a metal reacts with cold water, you get the metal hydroxide. However, the reaction soon stops because the magnesium hydroxide formed is almost insoluble in water and forms a barrier on the magnesium preventing further reaction. After several minutes, some bubbles of hydrogen form on its surface, and the coil of magnesium ribbon usually floats to the surface. Very clean magnesium ribbon has a very slight reaction with cold water. Magnesium burns in steam to produce white magnesium oxide and hydrogen gas. That means that textbook (or these days, web) statements about it never get checked, and are just passed on regardless of their accuracy. This means that few teachers will ever have handled it - I certainly haven't. 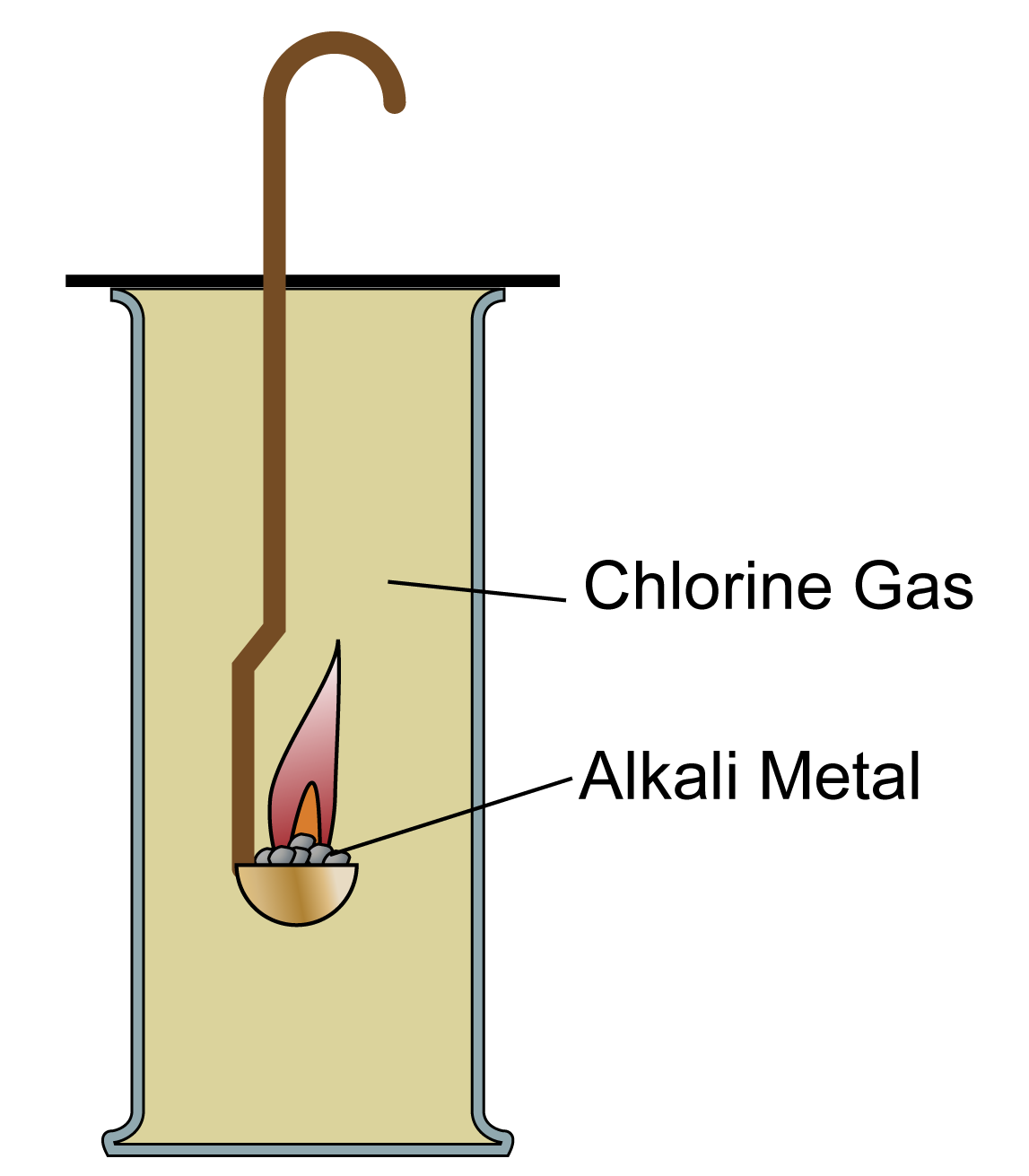
I think the problem is that beryllium is both expensive and carries major health risks. However, a researcher in the development of nuclear fusion pointed out to me that a lot of work is being done on the reaction between beryllium and steam because it could be a safety issue in reactor design, and sent me a photo showing the result of exposing beryllium to steam at 800☌. Note: Until November 2016, at this point I had the statement: "Beryllium has no reaction with water or steam even at red heat." This was commonly quoted in textbooks, and my sources were a degree level inorganic book, and a later A level inorganic book. (Equation just like the magnesium one below.) It uses these reactions to explore the trend in reactivity in Group 2.īeryllium reacts with steam at high temperatures (typically around 700☌ or more) to give white beryllium oxide and hydrogen. This page looks at the reactions of the Group 2 elements - beryllium, magnesium, calcium, strontium and barium - with water (or steam). REACTIONS OF THE GROUP 2 ELEMENTS WITH WATER Hydrogen chloride gas (from which we get hydrochloric acid) is an important industrial product.Reactions of the Group 2 elements with water There are four different oxides of the element. In addition to the ionic compounds that chlorine forms with metals, it also forms molecular compounds with non-metals such as sulfur and oxygen. Sodium hydroxide (also in high demand) is a by-product of the process. On an industrial scale, chlorine is produced by electrolysis of brines or even sea water. Small amounts of chlorine can be produced in the lab by oxidizing \(HCl\) with \(MnO_2\). Perhaps the best known compound of that type is sodium chloride, common table salt (NaCl). It forms -1 ions in ionic compounds with most metals. Like fluorine and the other members of the halogen family, chlorine is diatomic in nature, occurring as \(Cl_2\) rather than Cl. It forms strong ionic bonds with metal ions. Because chlorine is so highly reactive, it is found in nature in a combined state with other elements, such as NaCl (common salt) or KCl (sylvite). By itself, it is not combustible, but many of its reactions with different compounds are exothermic and produce heat.  
Chlorine is easily reduced, making it a good oxidation agent. \)Īt room temperature, pure chlorine is a yellow-green gas.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
